Azacitidine (Vidaza): Treatment for MDS and AML
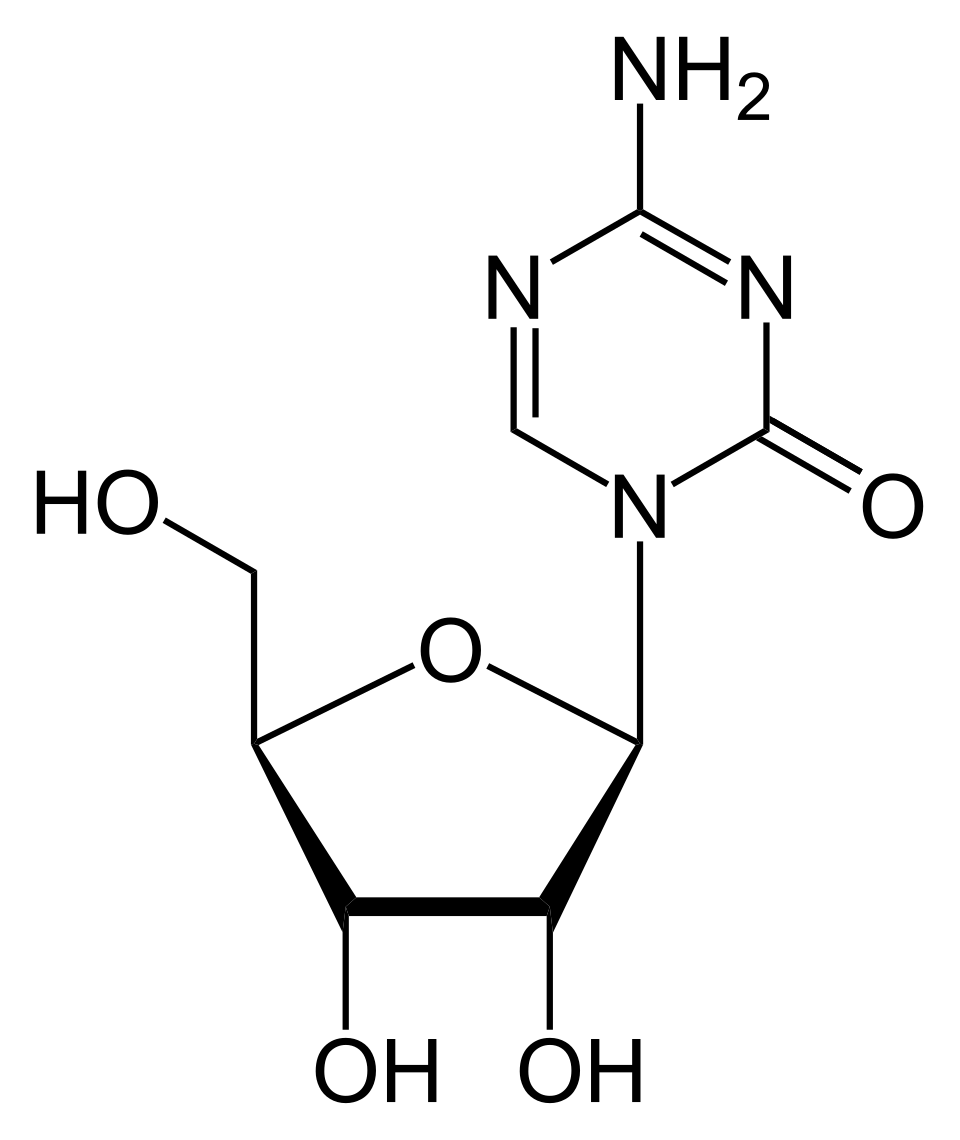
Azacitidine is an antineoplastic agent that offers a lifeline for patients with blood disorders where the bone marrow fails to produce healthy cells. It belongs to a class of drugs called hypomethylating agents. Its primary mechanism is to inhibit DNA methyltransferase, effectively "waking up" tumor suppressor genes that have been silenced by the disease, promoting normal cell maturation and inducing cancer cell death.
Azacitidine (Vidaza) is considered a standard of care for patients who are not eligible for hematopoietic stem cell transplantation. Unlike traditional high-dose chemotherapy, it works by epigenetic reprogramming of the cancer cells.
It is administered via subcutaneous injection (most common) or intravenous infusion.
Indications
- Myelodysplastic Syndromes (MDS): Treatment of adult patients with intermediate-2 and high-risk MDS according to the IPSS.
- Chronic Myelomonocytic Leukemia (CMML): For patients with 10-29% marrow blasts without myeloproliferative disorder.
- Acute Myeloid Leukemia (AML): Treatment of adult patients with 20-30% blasts and multi-lineage dysplasia, or elderly patients (>65 years) with >30% blasts who are not eligible for HSCT.
Dosage and administration
Treatment is given in cycles. Adherence to the schedule is vital.
- Cycle Schedule: Administered daily for 7 consecutive days, followed by a rest period of 21 days. The complete treatment cycle is 28 days.
- Dose: The recommended starting dose is 75 mg/m² of body surface area.
- Administration: Typically given as a subcutaneous injection (upper arm, thigh, or abdomen). Injection sites should be rotated. Pre-medication for nausea is often required.
- Duration: Patients should be treated for at least 6 cycles. Treatment continues as long as the patient benefits.
- Liver: Advanced malignant hepatic tumors.
- Pregnancy and Breastfeeding: The drug can cause fetal harm and should not be used during nursing.
- Known hypersensitivity to azacitidine or mannitol.
Side effects commonly affect blood counts and the digestive system.
- Hematologic: Thrombocytopenia (low platelets), neutropenia (low white blood cells), and anemia. Complete blood counts (CBC) are monitored frequently.
- Gastrointestinal: Nausea, vomiting, diarrhea, or constipation are very common. Anti-emetics are usually prescribed.
- Injection Site Reactions: Redness, bruising, or pain at the site of the subcutaneous injection.
- General: Fatigue, fever (pyrexia).
