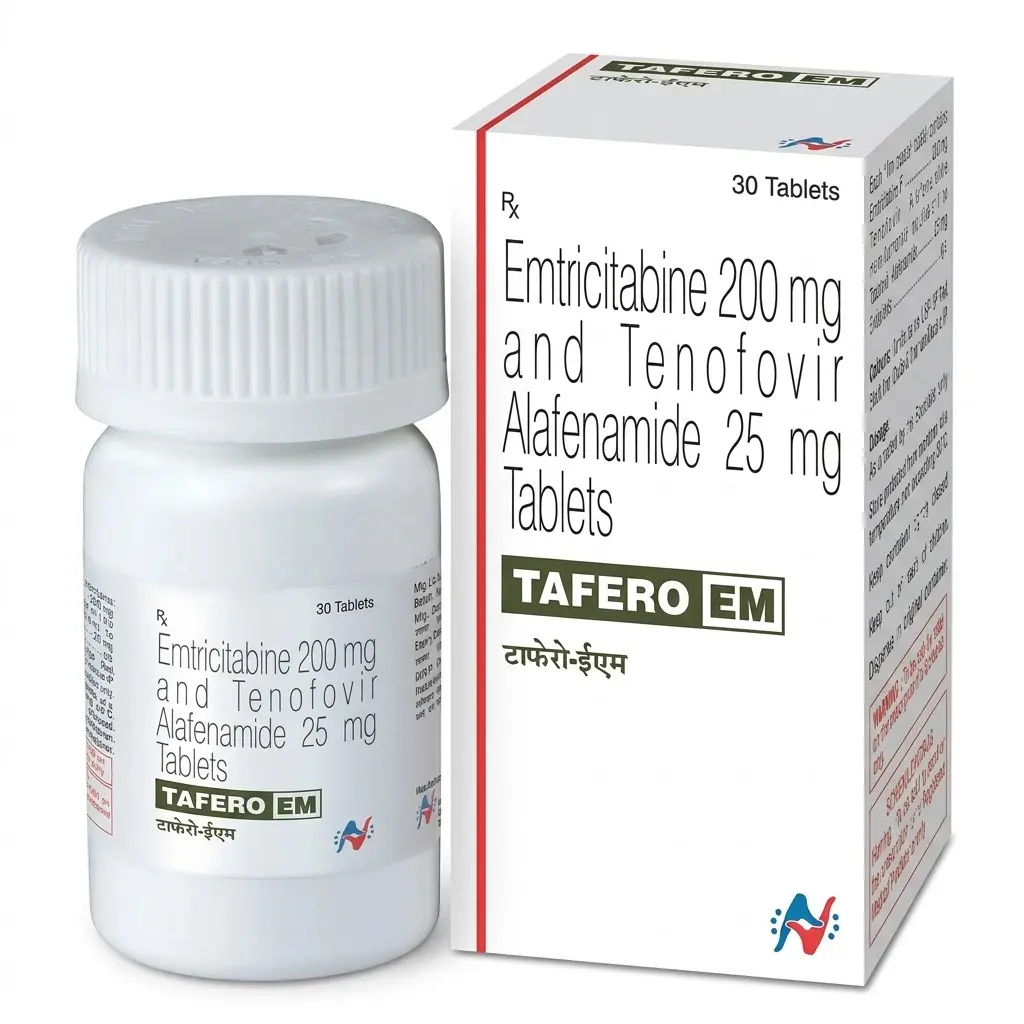
Tafero EM (Emtricitabine 200 mg / Tenofovir Alafenamide 25 mg)
Tafero EM is a next-generation medication for HIV treatment and prevention (PrEP). Active ingredients: Emtricitabine (200 mg) + Tenofovir Alafenamide (25 mg).
Manufacturer: Hetero Healthcare (India). It is the generic equivalent of the advanced drug Descovy.
The Safety Revolution:
Unlike older drugs like Truvada (TDF), Tafero EM uses Tenofovir Alafenamide (TAF). This molecule targets cells more efficiently, allowing for a much lower dose (25 mg). The result is a highly effective drug with a superior safety profile for kidneys and bone density.
✅ Top Choice: Considered the modern gold standard for long-term therapy and prophylaxis.
Approved for:
- HIV-1 Treatment: In combination with other antiretroviral agents (e.g., Dolutegravir) for adults and pediatric patients.
- PrEP (Pre-Exposure Prophylaxis): To reduce the risk of sexually acquired HIV-1 infection in at-risk adults and adolescents.
Standard Dosage: One tablet taken orally once daily.
⚠️ Administration Guide:
- Take at the same time every day to maintain viral suppression or protection.
- Can be taken with or without food.
- For PrEP users: Adherence is crucial. Missing doses significantly increases the risk of HIV infection.
- Unknown HIV Status (for PrEP): You must test negative for HIV immediately before starting PrEP. Taking this drug alone while HIV-positive can lead to drug resistance.
- Severe hepatic impairment.
- Hypersensitivity to Emtricitabine or Tenofovir.
Tafero EM is generally well-tolerated with fewer side effects than older TDF-based drugs:
- Mild nausea or headache.
- Renal & Bone Safety: Significantly lower risk of kidney toxicity and osteoporosis compared to Truvada/Tenvir-EM.
- Potential mild weight gain.
Similar products
What Customers Say
No reviews yet
Your review can be the first!