Ceritinib (Zykadia): ALK Inhibitor for NSCLC
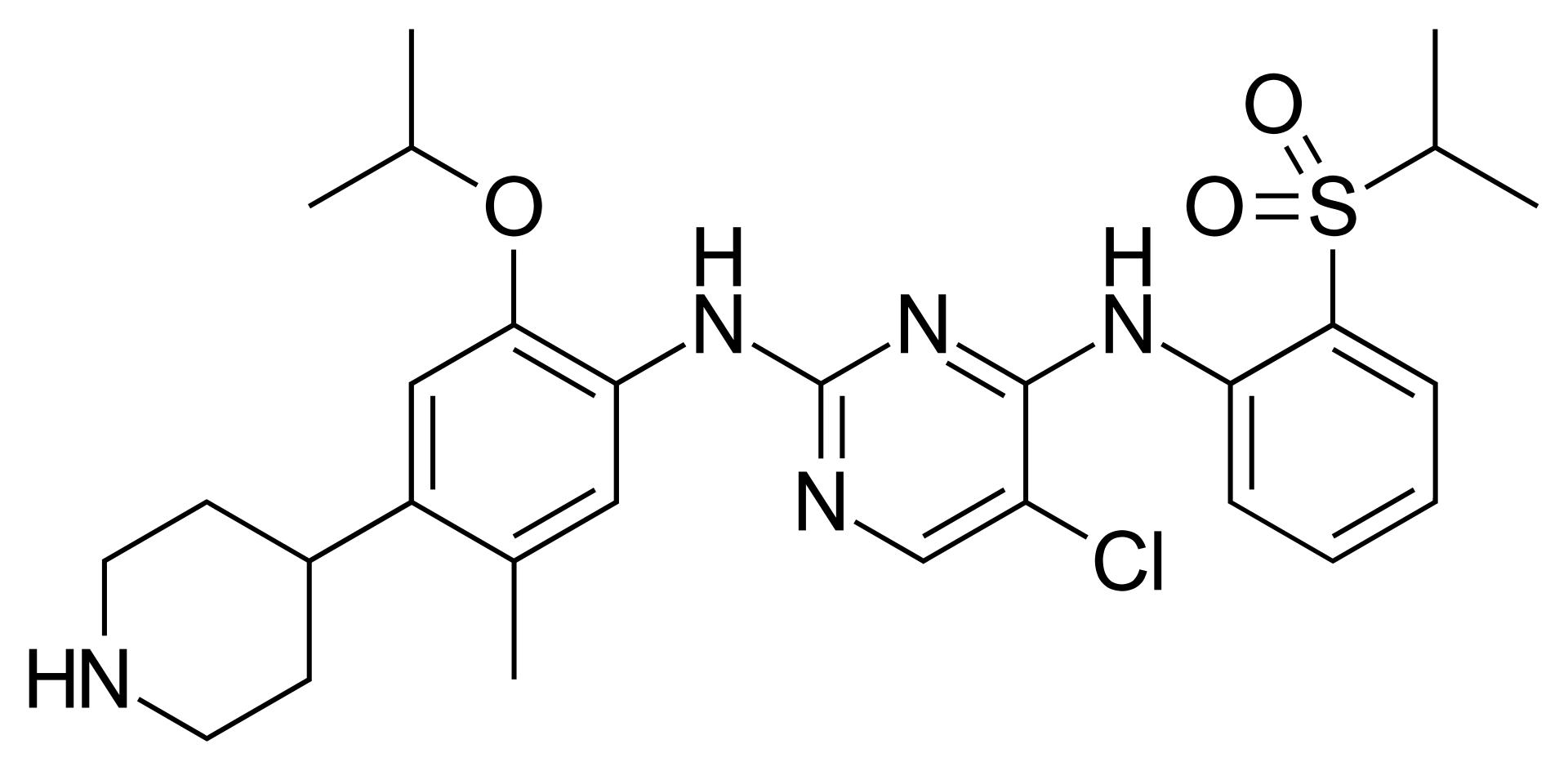
Ceritinib is a highly potent, oral tyrosine kinase inhibitor (TKI) that targets Anaplastic Lymphoma Kinase (ALK). It is a targeted therapy designed specifically for a subset of lung cancer patients whose tumors are driven by the ALK gene rearrangement.
Ceritinib (Zykadia) works by blocking the defective ALK protein, thereby cutting off the signal that tells cancer cells to grow and divide. It is known to be more potent than first-generation inhibitors like crizotinib and can be effective even if the cancer has developed resistance to prior therapies.
To improve patient comfort and reduce gastrointestinal toxicity, the dosing regimen has been optimized to be taken with food.
Indications
- ALK-positive NSCLC: Treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) whose tumors are anaplastic lymphoma kinase (ALK)-positive.
- Line of Therapy: Indicated both as a first-line treatment and for patients who have progressed on or are intolerant to crizotinib.
Dosage and administration
Strict adherence to food intake instructions is required to manage side effects.
- Recommended Dose: 450 mg orally once daily. (Usually three 150 mg capsules).
- Administration: Must be taken with food. Taking it with a meal significantly improves gastrointestinal tolerability compared to the older fasting regimen.
- Swallowing: Swallow capsules whole with water. Do not chew or crush.
- Cardiac Conditions: Congenital Long QT syndrome.
- Pregnancy and Breastfeeding: Can cause fetal harm. Effective contraception is required during treatment.
- Severe hypersensitivity to ceritinib.
Monitoring for gastrointestinal, hepatic, and metabolic effects is standard.
- Gastrointestinal: Diarrhea, nausea, vomiting, and abdominal pain are very common.
- Hepatotoxicity: Elevations in liver enzymes (ALT/AST). Monthly blood tests are recommended.
- Hyperglycemia: Increased blood sugar levels.
- Cardiac: Bradycardia (slow heart rate) and QT interval prolongation.