Capmatinib: MET Inhibitor for Non-Small Cell Lung Ca
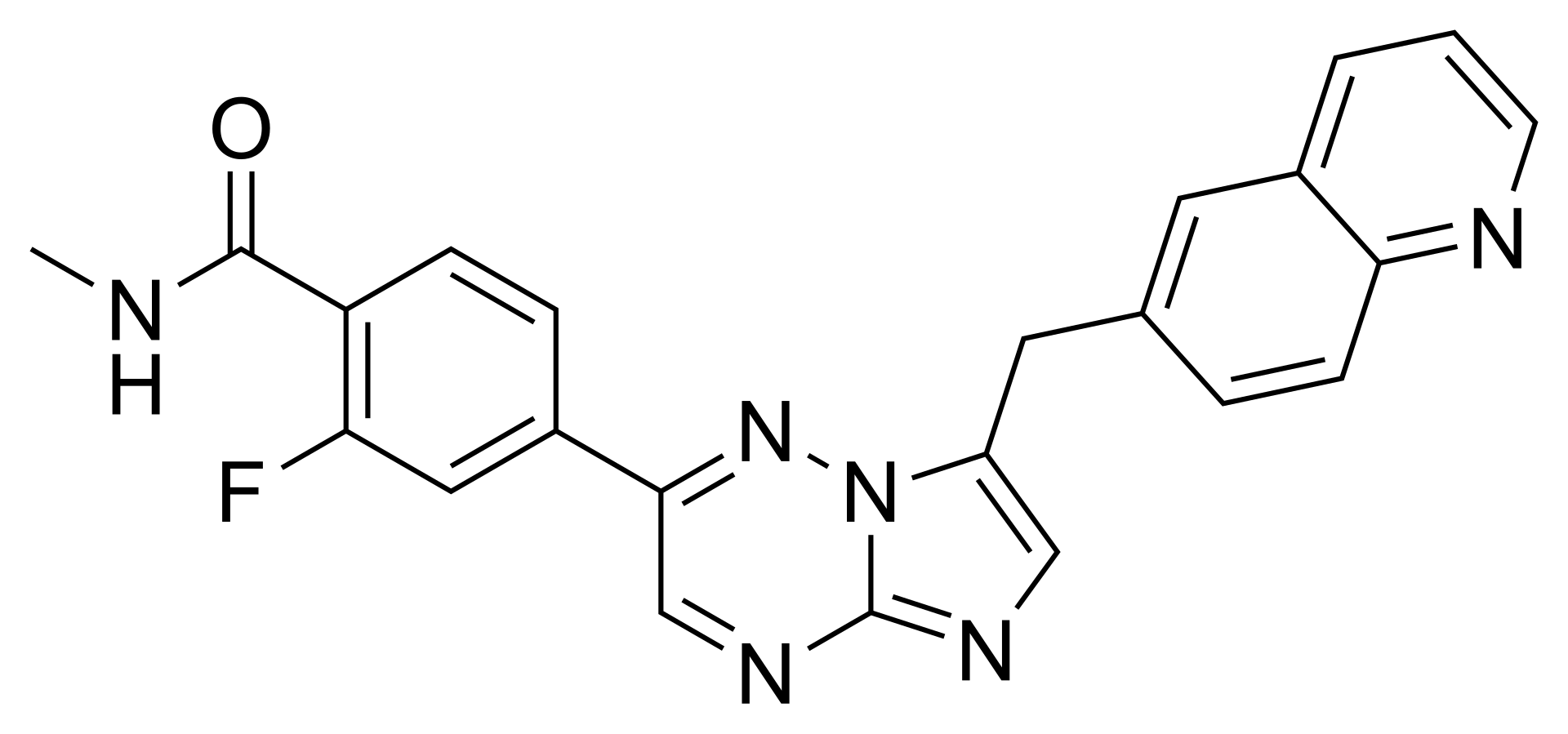
Capmatinib is a highly selective, oral inhibitor of the MET receptor tyrosine kinase (mesenchymal-epithelial transition factor). The drug blocks the MET receptor, suppressing intracellular signaling pathways responsible for tumor cell growth, survival, and metastasis.
Capmatinib demonstrates the greatest efficacy in patients with METex14 skipping mutations — a recognized oncogenic driver in non-small cell lung cancer. The drug is also active in tumors with MET gene amplification. On Unifarm, you can find generic versions of medications (such as Tabrecta) containing this active ingredient.
Indications
- Non-Small Cell Lung Cancer (NSCLC) with METex14 Skipping Mutation: Treatment of adult patients with metastatic NSCLC whose tumor harbors a mutation leading to MET exon 14 skipping, as detected by a validated test.
- First-line therapy: For patients with newly diagnosed metastatic NSCLC with METex14 skipping mutation.
- Previously treated patients: For patients who have received at least one prior line of systemic therapy.
Dosage and administration
Available as film-coated tablets for oral administration.
Standard Regimen:
- Recommended Dose: 400 mg taken orally twice daily approximately 12 hours apart.
- Administration: Swallow tablets whole with water. May be taken with or without food. Do not crush, chew, or break tablets.
- Dose Reduction: For adverse reactions, the dose may be reduced to 300 mg twice daily, then to 200 mg twice daily.
Treatment should continue until disease progression or unacceptable toxicity. Regular monitoring of hepatic function and assessment for interstitial lung disease (ILD) is recommended. No dose adjustment is needed with strong CYP3A4 inhibitors; however, concomitant use with strong CYP3A4 inducers should be avoided.
- Hypersensitivity to capmatinib or any component of the product.
- Pregnancy and breastfeeding (can cause fetal harm).
- Severe hepatic impairment (use not studied).
- Active interstitial lung disease (ILD) / pneumonitis.
Capmatinib therapy requires regular monitoring of hepatic, renal, and pulmonary function. Common adverse reactions include:
- Gastrointestinal: Nausea (very common), vomiting, diarrhea, constipation, decreased appetite.
- Hepatic: Elevated transaminases (ALT, AST), increased bilirubin, elevated alkaline phosphatase — hepatotoxicity requiring regular monitoring.
- Renal: Increased blood creatinine, peripheral edema.
- Pulmonary: Interstitial lung disease (ILD) / pneumonitis — serious adverse effect; treatment must be immediately discontinued if symptoms (dyspnea, cough, fever) appear.
- General: Fatigue, peripheral edema, weight loss.
- Hematologic: Lymphopenia, leukopenia.
- Musculoskeletal: Back pain, arthralgia, myalgia.
- Dermatologic: Photosensitivity (prolonged sun exposure should be avoided).