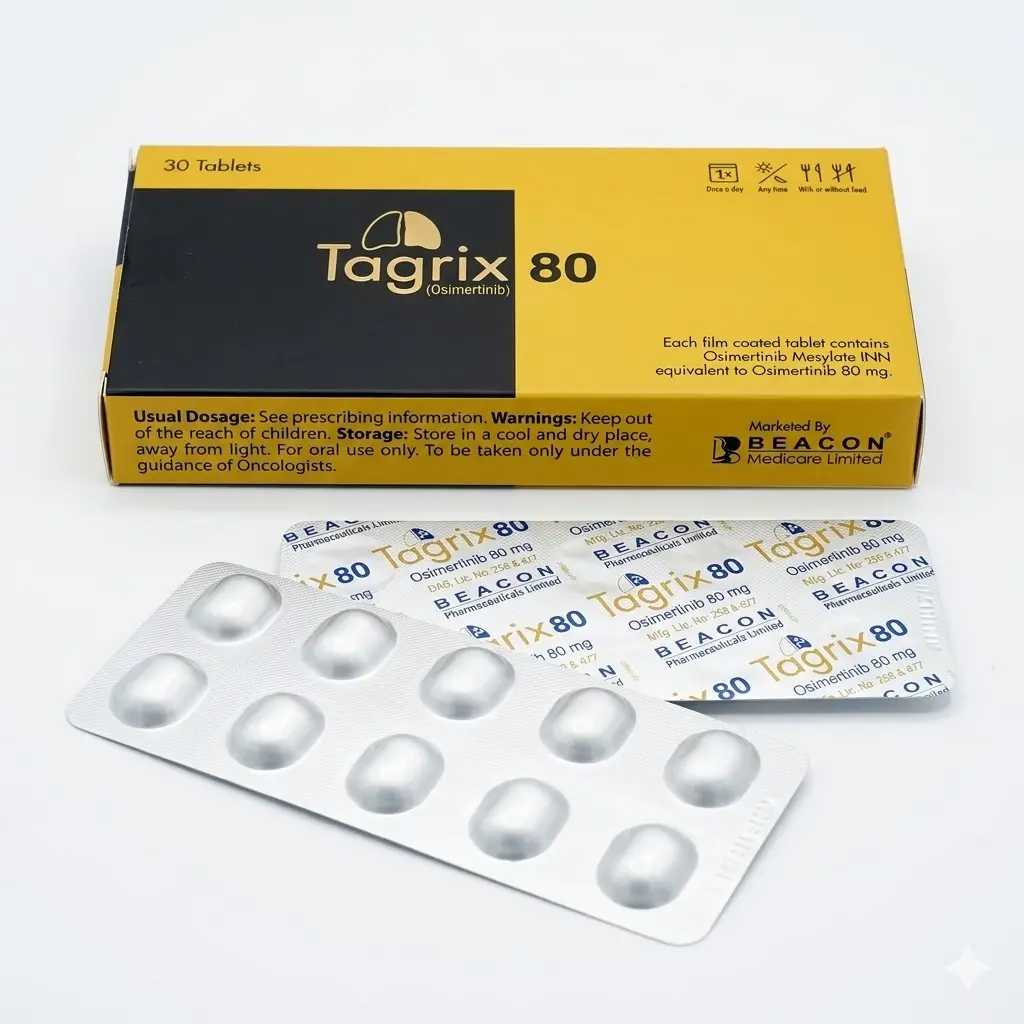
Tagrix 80 (Osimertinib) 80 mg Tablets
Tagrix 80 mg (Osimertinib) is a third-generation EGFR tyrosine kinase inhibitor (TKI) used for targeted lung cancer therapy.
Manufacturer: Beacon Medicare Limited. Each pack contains 30 tablets. It is designed to target specific genetic mutations, including the T790M resistance mutation, and is highly effective at crossing the blood-brain barrier to treat brain metastases.
Key Feature:
Considered the standard of care for EGFR-mutated NSCLC due to its superior efficacy and safety profile compared to earlier generation inhibitors.
Indicated for the treatment of adult patients with Non-Small Cell Lung Cancer (NSCLC):
- 🔹 First-line treatment: For patients with metastatic NSCLC whose tumors have EGFR exon 19 deletions or exon 21 L858R mutations.
- 🔹 Second-line treatment: For patients with EGFR T790M mutation-positive NSCLC who have progressed on prior EGFR-TKI therapy.
- 🔹 Adjuvant therapy: After tumor resection.
Packaging: 80 mg tablets.
⚠️ ADMINISTRATION PROTOCOL:
- Recommended Dose: 80 mg orally once daily.
- Food Intake: Can be taken with or without food.
- Timing: Take at the same time each day.
- Administration: Swallow whole. If unable to swallow, the tablet may be dispersed in 50 ml of non-carbonated water (do not crush).
- Supply: One pack (30 tablets) provides a full 30-day supply.
- Hypersensitivity to Osimertinib.
- Pregnancy and breastfeeding (embryo-fetal toxicity).
- Active Interstitial Lung Disease (ILD) or pneumonitis.
- QTc interval prolongation.
Common adverse reactions include:
- 🩸 Dermatologic: Rash, dry skin, paronychia (nail infection/inflammation), pruritus.
- 📉 Gastrointestinal: Diarrhea (very common), stomatitis (mouth sores).
- 🦠 Blood: Decreased leukocyte or platelet counts.
- ⚠️ Cardiac: QT prolongation (ECG monitoring is recommended).
Similar products
What Customers Say
No reviews yet
Your review can be the first!