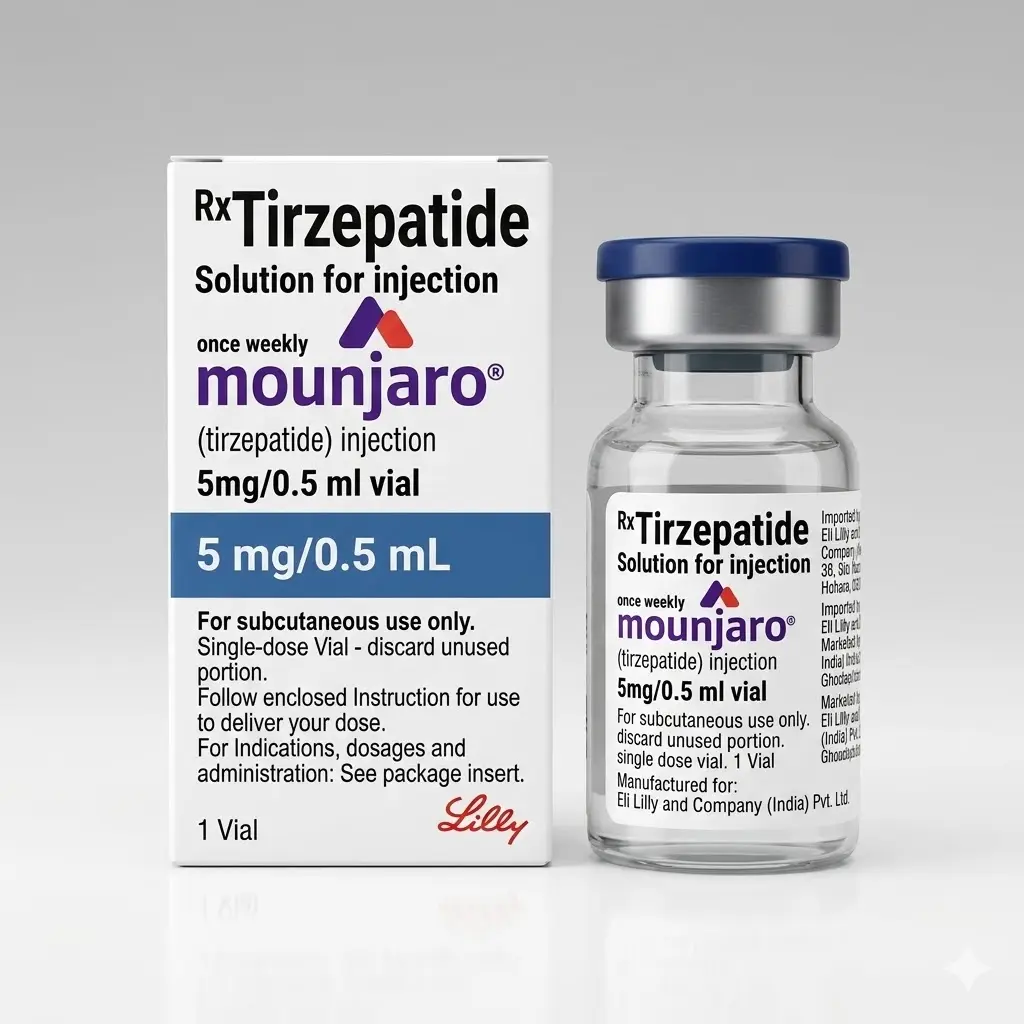
Mounjaro 5 mg (Tirzepatide) 5 mg/0.5 mL
Mounjaro 5 mg (Tirzepatide 5 mg) is the first therapeutic maintenance dose of the dual GIP/GLP-1 receptor agonist.
Manufacturer: Eli Lilly (India). This 5 mg vial represents Step 2 in the dosage escalation protocol. Unlike the 2.5 mg starter dose, the 5 mg dose is considered clinically effective for glycemic control and initiates more significant weight loss results.
Mechanism of Action ("Metabolic Reset"):
By activating both GIP and GLP-1 receptors, Tirzepatide 5 mg provides a stronger signal to the brain to stop eating and increases insulin sensitivity significantly. It effectively lowers fasting and post-meal glucose levels while promoting substantial calorie deficit through appetite suppression.
✅ Key Benefit: Marked improvement in HbA1c levels and accelerated body weight reduction compared to the starting dose.
Indicated for adults who have completed the initial 4-week course of 2.5 mg:
- 🍬 Type 2 Diabetes Mellitus: To improve glycemic control.
- ⚖️ Chronic Weight Management: For obesity or overweight individuals with weight-related comorbidities.
Packaging: Single-dose Vial (5 mg / 0.5 ml).
⚠️ DOSAGE PROTOCOL (Step 2):
- 💉 5 mg Dose: Inject subcutaneously once weekly. Typically prescribed for weeks 5-8 of treatment.
- 🛑 Warning: DO NOT start therapy with 5 mg. Patients must complete 4 weeks on 2.5 mg first to mitigate severe gastrointestinal distress.
- 📍 Storage: Keep refrigerated at 2°C to 8°C (36°F to 46°F).
- Personal or family history of Medullary Thyroid Carcinoma (MTC).
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
- Pregnancy and breastfeeding.
- Severe gastrointestinal disease (e.g., gastroparesis).
Side effects may re-emerge during dose escalation:
- 🤢 Gastrointestinal: Nausea, vomiting, diarrhea, constipation (usually transient).
- ⚠️ Pancreatitis: Risk of inflammation of the pancreas.
- Hypoglycemia (if used with Sulfonylureas or Insulin).
Similar products
What Customers Say
No reviews yet
Your review can be the first!